INTRODUCTION
In 1984 Marshall and Warren[1] identified Helicobacter pylori (H. pylori) from gastric biopsy culture. In 1994, H. pylori was recognized as definite carcinogen by International agency for research on cancer. H. pylori induced gastric cancer (GC) is accountable for 5.5% of global cancer burden[2].
H. pylori is spiral shaped, gram-negative, microaerophilic, flagellated human pathogen that successfully colonizes gastric mucosa of majority of individuals[3]. Epidemiologically, the H. pylori infection is exists all over the world, but colonization rates vary considerably; high in developing compared to the developed world[4]. H. pylori acquisition thought to occurs in early childhood. Fecal-oral or oral-oral were considered as possible route of H. pylori transmission[4,5]. H. pylori urease is among the various virulence factors that aids in colonizing the highly acidic environment of stomach via breakdown of urea into ammonia, generating hospitable locale for its colonization[6] (Figure 1). Among the majority of H. pylori infected individuals only a small percentage of colonized individuals develop severe clinical disease such as GC. Determing factors responsible for variation in clinical outcomes of H. pylori infection are still not well studied. For a longer period of time association between H. pylori and GC was debatable. A study from Japan on 1526 patients gives a clear evidence that H. pylori infection is significantly associated with risk of developing GC[6]. Proof that H. pylori has an influence on early stages of gastric carcinogenesis is demonstrated by randomized prospective studies which shows association between H. pylori eradication and reduction of premalignant tumors[7,8]. Research on experimentally challenged Mongolian gerbils, provide evidence concerning H. pylori eradication with attenuation of developmental process related to GC progression[9,10]. Together these studies authenticate that H. pylori plays a key role in development of GC and indicate that H. pylori eradication provide protection against H. pylori-induced GC. Interaction among environmental factors, host genetic polymorphism and bacterial virulence attributes collectively influence the clinical outcome of H. pylori infections[11].
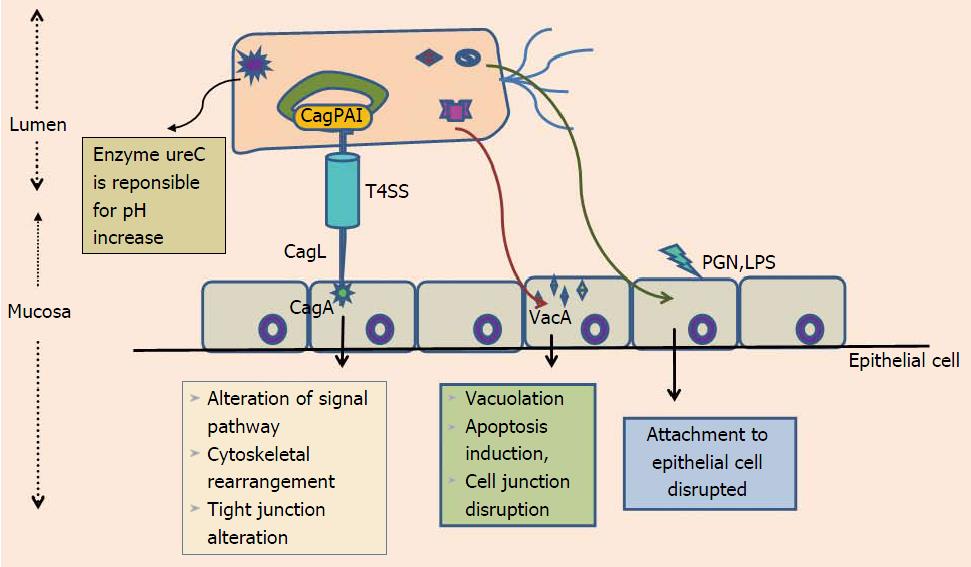
Figure 1 Interaction between Helicobaceter pylori type IV secretion system and virulence determinants such as CagA, CagL, lipopolysaccharides, pepetidoglycan and vacuolating cytotoxin gene, with mucosal epithelial cells, resulting in alteration of signal pathways, cell polarity disruption and vaccuolation, which ultimately leads to death.
T4SS: Type IV secretion system; LPS: Lipopolysaccharides; PGN: Pepetidoglycan; VacA: Vacuolating cytotoxin; CagPAI: Cag pathogenicity island.
This review aims to highlight recent progress in H. pylori pathogenesis, especially the bacterial and host factors that are involved in the host-pathogen interaction during persistent colonization. It also highlights the host immune response towards H. pylori colonization and its effect on diverse clinical outcomes, especially on advancement leading to GC.
EPIDEMIOLOGY OF GC
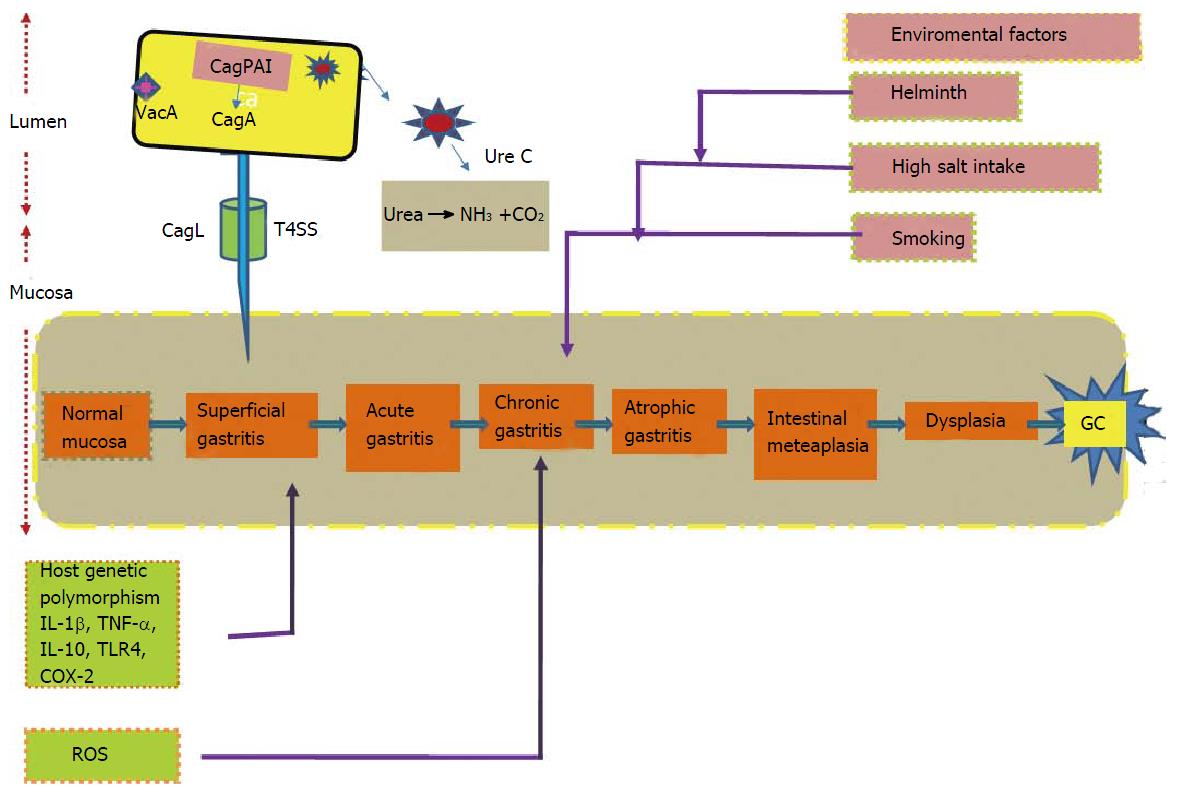
GC is a multifactorial disease. Correa’s model describes array of event beginning from chronic active gastritis, atrophic gastritis, intestinal metaplasia, dysplasia and eventually leads to GC[12] (Figure 2). Risk factors for the development of the GC include interaction among the pathogen, environmental and host-related factors[13]. World Health Organization recognized H. pylori as class I carcinogen in 1994. GC is identified as the fifth most common malignancy and third leading cause of cancer-related morbidity globally, constituting 9.7% of all cancer-related mortality[14]. Highest age-standardized mortality rate (ASMR) is predicated for Eastern Asia (28.1 per 100000 in men, 13.0 per 100000 in women), the lowest ASMR in North America (2.8 and 1.5 per 100000, respectively)[15]. Studies reported high mortality rates are from East Asia, Central and Eastern Europe, Central and South America[15]. Developing countries have high burden of GC compared to the developed world and GC accounts for approximately 70% of both new cases and deaths[16]. Categorizing on basis of gender, 466900 cases of males were reported from developing as compared to 173700 cases from developed countries and for females the corresponding disease load was 247000 and 102000 cases, respectively. GC is associated with age incidence; commonly occurs in age group of 55 to 80 years, rare among young individual. Frequency of GC rates are two fold higher in males than females[17].
Figure 2 Combination of host, bacterial and environmental factors, which act in a synergetic way, resulting in development of precancerous cascade that ultimately leads to development of gastric cancer.
ZO-1: Zonula occludens 1; CagPAI: Cag pathogenicity island; JAM: Junctional adhesion molecule; VacA: Vacuolating cytotoxin; Cytc: Cytochrome.
Over past decades in western nations, GC has considerably declined. The possible reasons behind this reduction include fall in H. pylori prevalence accompanied by better hygienic practices and innovative medical diagnostic facility. Despite the decline in GC incidence in developed world, the scenario of developing world is diverse. GC incidence and mortality rate remain very high in the developing nations, particularly in regions of East Asia and South America[17]. It is expected that if appropriate measures are not implemented the number of estimated GC cases are likely to increase in future.
PATHOLOGICAL DIFFERENTIATION
Majority of gastric malignant tumors are adenocarcinomas. Histologically Lauren categorized gastric adenocarcinoma in intestinal and diffuse subtypes. Intestinal type adenocarcinoma is event dependent, start from chronic atrophic gastritis to intestinal metaplasia to dysplasia and finally carcinoma. Intestinal type adenocarcinoma is more frequent in developing world, common in male, and associated with age incidence, whereas diffuse type occurs more often in younger patients having family history of cancers, more frequent in females, background of atrophic gastritis is not prerequisite condition for its occurrence[18,19]. Anatomical site of origin is another way of differentiation of gastric adenocarcinoma. Tumors arising in the cardia region of the stomach are said to be proximal, and those from body and antrum (non-cardia region) as distal. Histological subtypes represent etiological and epidemiological differences between the two tumor sub sites. Globally GC incidence is declining. However, studies show rise in incidence of cardia carcinoma which may be partly due to more accurate reporting and fall in incidence of distal cancers[20].
H. PYLORI AS A RISK FACTOR FOR GC
Colonization of the stomach by H. pylori causes development of gastritis. H. pylori is truly an “opportunistic” bacterium that uses various well defined virulence factors as tool for attachment and persistent colonization of human gastric mucosa. The possible transmission route is fecal-oral, but contaminated food or water are also reported[21,22]. The most likely sources are person-to-person contact in families and/or exposure to a common source of infection such as contaminated water or food as supported by majority of data[23]. This notion is supported by studies of children in custodial care where the prevalence of infection is higher than expected and from studies of crowded families in which there is at least one infected child[24].
Before attachment of H. pylori to gastric epithelium, it has to first cross the thick mucus layer by adhering to the mucosal surface. This is aided by the presence of unipolar sheathed flagella, which allows H. pylori to quickly move from inhospitable low pH of gastric lumen to surface epithelium where pH is high and favorable for its successful colonization despites efforts made by the host to get rid of this bacterium. Non-motile mutant H. pylori strains fail to colonize the stomach of gnotobiotic piglets[25,26]. In majority of infected individuals colonization results in development of inflammatory and immune responses against H. pylori, but in some subjects H. pylori infection becomes chronic and leads to induction of gastric inflammation which can eventually lead to destruction of normal gastric glands and their replacement by intestinal-type epithelium resulting in atrophy of gastric mucosa.
The risk for atrophic gastritis depends on pattern as well as extent of distribution of chronic active inflammation. The individuals with lower acid output show a higher tendency towards atrophy[27]. Reduction in gland size and level of intestinal metaplasia were associated with rise in GC risk by 5- to 90-folds depending on the extent and severity of atrophy[28].
Increased odds ratios were evident from case-control studies that aimed to seriously study the signs of earlier H. pylori infection in GC patients and controls for development of non-cardia GC in presence of H. pylori infection[29]. This fact is supported by data from animal models including Mongolian gerbil model, in which H. pylori infection induces atrophic gastritis and GC[30-32]. A small number of subjects for research purposes were deliberately infected with pathogenic H. pylori strain and individuals developed acute inflammation of gastric mucosa with neutrophilic infiltration[33,34]. Volunteers after several decades when exposed repeatedly to intragastric pH-electrodes contaminated with H. pylori developed conditions called “epidemic hypochlorhydria”[34]. Such hypochlorhydric gastritis can either resolve spontaneously or change into chronic gastritis.
ROLE OF HOST GENETICS
H. pylori infection results in three possible outcomes. First is corpus-predominant gastritis beginning from atrophic gastritis to hypochlorhydria and finally to GC. Second type results in a pangastritis having slightest impact on the host gastric acid production. Duodenal ulcer is third outcome, where an antrum-predominant gastritis leads to hyperchlorhydria. There arises controversy that infections of H. pylori can predispose to two equally exclusive situations. The possible explanation why some people are more expected to develop GC phenotype when compared with others may be due to disparity among individual host response to H. pylori infections (Figure 1). Initial evidence for the importance of host genetic polymorphisms was reflected in the study where a rise in incidence of atrophic gastritis and hypochlorhydria was evident from relatives of H. pylori induced GC patients than controls[35].
Pro-inflammatory cytokine like interleukin-1beta (IL-1β) act as a powerful negative regulator of acid secretion. IL-1β gene is now considered as a potential contender for host genetic polymorphisms that may elevates GC risk. Individuals possessing IL-1β gene cluster polymorphisms have 2–3-folds increased risk of non-cardia cancer[36,37] (Figure 2). Elevated levels of TNF-α in gastric mucosa of H. pylori infected individuals were evident from numerous studies. However, down regulation of anti-inflammatory cytokine IL-10, that suppresses the level of pro-inflammatory cytokines including IL-1β, TNF-α and interferon-γ (IFN-γ) is also reported[36].
The risk associated with GC development in H. pylori infected individuals upsurges 27-folds in individuals with three or four polymorphisms[38]. This evidently illustrates that interaction between host genetics and environment plays a key role in progression of GC, by regulating hosts adaptive immune response resulting in transformation of normal gastric mucosa to neoplastic one.
IL-8
Higher expression of chemokine IL-8 and polymorphism (promoter region) has been reported in studies and linked with increased risk for GC[39]. Study on Caucasian populations proved that relationship among functional polymorphism within Toll like receptor 4, risk of GC and decrease in production of anti-inflammatory cytokine IL-10[40]. These studies reflect that host genetic polymorphisms are capable of modulating the innate immune response which results, severe inflammation and premalignant lesions in H. pylori infected individuals (Figure 2). These studies raises a query that whether H. pylori strain characteristics are responsible for increasing cancer risk employed by host genotypes, needs to be studied further. Odds ratios for non-cardia GC were highest for individuals with elevated IL-1β expression, colonized by H. pylori vacAs1-type strains[41].
It is evident from case-control studies that H. pylori successfully form a vital equations with host by its ability to send and receive signals from its hosts[42,43]. Only certain H. pylori strains enhance the possibility of carcinogenesis because the equilibrium is likely different for each colonized individual. For example, individual infected with CagA strains leads to severe gastritis, which results in rise of proinflammatory cytokines levels that are responsible for both amplifying the mucosal inflammatory response as well as reducing the acid production. This creates a milieu encouraging growth of H. pylori that promote inflammation and continually produce oxidative stress, thus augmenting risk for transformation of normal mucosa to neoplastic through series of events (Figure 2).
Cyclooxygenase
H. pylori triggers numerous forms of proinflammatory cyclooxygenase (COX) enzymes. Production of endoperoxide from arachidonic acid is brought by COX enzymes. Enzymes prostaglandin synthases produces prostaglandins and various eicosanoids from endoperoxide[44]. Important role is played by prostaglandins in regulating physiologic processes for instance immunity and development. Two COX isoforms (COX-1 and COX-2) have been categorized on the basis of variances in expression characteristics and inhibition profiles for nonsteroidal anti-inflammatory drugs (NSAIDs). COX-2 expression is inducible while COX-1 is constitutively expressed in cells and tissues[45-47]. Expression of COX-2 can be stimulated by proinflammatory cytokines, growth factors such as TNF-α, IFN-γ and IL-1. COX-2 expression are raised in H. pylori infected human gastric mucosa, gastric premalignant and malignant lesions[47-49]. Inhibitors of COX (aspirin and NSAIDs) are associated with reduced risk of non-cardia GC[50]. Numerous studies demonstrate substantial role of COX-2-generated products involved in promoting neoplasia. Mechanisms like apoptosis inhibition, regulation of expression of cell surface adhesion, and production of promoting factors of neoplasia leads to malignancy[51,52] (Figure 2).
H. PYLORI VIRULENCE FACTORS
Cag pathogenicity island
H. pylori have genetically heterogeneous genome. A number of H. pylori virulence factors are supposed to play an essential role in diverse clinical outcome of H. pylori infections. The Cag pathogenicity island (CagPAI) is a 40-kb region, consisting of 32 genes, flanked by 31-bp direct repeats. CagPAI is an island consisting of virulence genes, which are acquired by horizontal transfer. CagPAI encodes a type IV secretion system (T4SS) that is responsible for the entrance of a most remarkably investigated H. pylori virulence determinant effector protein CagA[53-55] (Figure 1). Positive association of CagA was found with peptic ulcer disease[56,57]. Due to its association with several gastroduodenal pathologies, initially CagA was considered as an indicator for presence of the entire CagPAI but as research speeded up, studies demonstrated that despite its presence, CagPAI intactness and clinical outcome varied.
More or less 70% of H. pylori strains from western world and nearly 100% of East Asian strains express virulent protein CagA[54,58,59]. Majority of H. pylori strains induces superficial gastritis but the risk for chronic gastritis, atrophic gastritis, metaplasia, and non-cardia GC with intact CagPAI is much higher compared to those that lacked it[56,57,60-66]. Among 32 genes of CagPAI, 18 genes are thought to code for structural parts of a T4SS, this system is responsible for exporting peptidoglycans and cagA into host gastric epithelial cells, via forming a pilus like assembly connecting bacterial and host epithelial membrane (Figure 1).
CagA
CagA is terminal gene product of the CagPAI. Classifying H. pylori strains on the basis of presence and absence of cagA into cagA-positive and cagA-negative strains. After the H. pylori attachment to epithelial cell, CagA is internalized through T4SS apparatus. After translocation, CagA is tyrosine phosphorylated at glutamate-proline-isoleucine-tyrosine-alanine (EPIYA) motif, i.e., EPIYA motif which is associated with cell morphological changes known as “the hummingbird phenotype,” which results in increased cellular migration[67-71].
Polymorphic region of CagA, has been identified within the carboxy-terminal and distinguished by different amino acid sequences. Till date, four distinct EPIYA motifs (EPIYA-A, -B, -C and D) are known[72,73]. EPIYA-A and -B motifs are present in strains all over the world, whereas EPIYA-C is specific to western world (Europe, North America, and Australia). Variation in number of EPIYA-C sites occurs, while majority of CagA proteins contain a single EPIYA-C site (A-B-C type). The level of phosphorylation of EPIYA-C sites is greater than EPIYA-A and EPIYA-B sites. Risk for development of GC is found to be associated with the number of cagA EPIYA-C in western strains[74]. EPIYA-D motif is exclusive to East Asian strains (from Japan, South Korea, and China), and strains possessing this motif produces higher level of IL-8 from gastric epithelial cells as compared to strains harboring western A-B-C-type CagA[72,75].
CagA phosphorylation-dependent host cell signaling
Kinase families of Abl and Src are responsible for phosphorylation of CagA into phospho-CagA. Interaction between phosphorylated CagA and various intracellular effectors, triggers an eukaryotic tyrosine phosphatase (SHP-2), which results in continuous stimulation of extracellular signal-regulated kinases 1 and 2 (ERK1/2), Crk adaptor[76] and C-terminal Src kinase in a tyrosine phosphorylation-dependent manner. In East Asian A-B-D types, negative response is induced by interactions of phospho-CagA with C-terminal Src kinase resulting down regulation of Src signaling[77] (Figure 3).
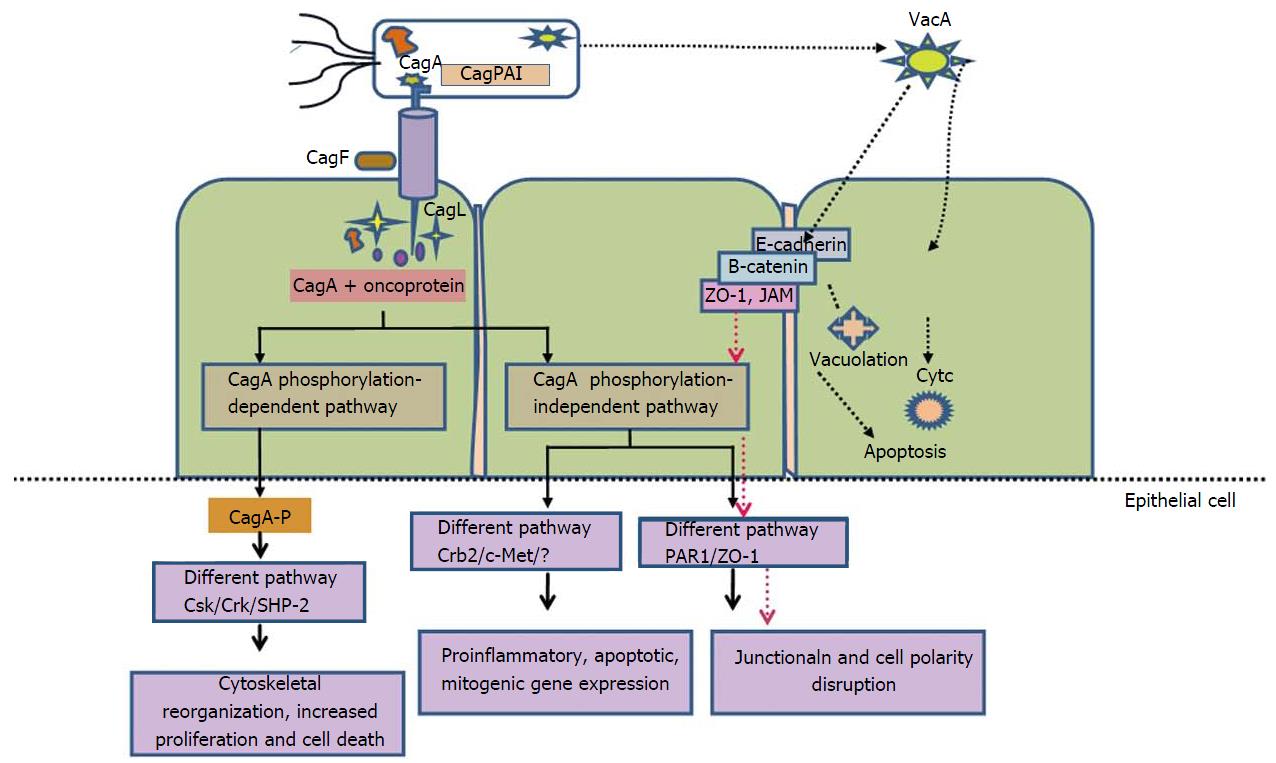
Figure 3 Schematic representation of multiple pathways of Helicobacter pylori pathogenesis involved type IV secretion system and internalization of virulence determinants like CagA and oncoproteins; CagA-phosphorylation dependent and CagA-phosphorylation independent pathways leads to cytoskeletal reorganization, increase proinflammatory and mitogenic gene expression.
Another major virulent factor, VacA is responsible for alteration of junction and cell polarity by binding with tight junction molecules such as E-cadherin, ZO. VacA also causes mitochondrial membranes depolarization, Cytc release from mitochondria to cytosol and caspase-3 activation followed by cell apoptosis. T4SS: Type IV secretion system; VacA: Vacuolating cytotoxin; ZO: Zonaoccludans; Cytc: Cytochrome; H. pylori: Helicobacter pylori.
Experimental studies on cell lines revealed that CagA internalization give rise to “hummingbird phenotype”. These alterations are characterized by cell elongation and cell scattering[69,78]. Additional study also indicates that interplay among phosohorylated CagA, dephosphorylation of SHP-2 and down-regulation of focal adhesion kinase, causes cell elongation[69,79]. A different mechanism of cell elongation by phosphorylated CagA is by making a defect in cell retraction; yet the signaling molecules prerequisite for this phenotype remain vague[80]. Phosphorylated CagA obstructs the enzymatic activity of c-Src, which leads to tyrosine dephosphorylation of actin binding proteins such as cortactin, ezrin, and vinculin, ultimately results in cell elongation[81-83] (Figure 3).
CagA phosphorylation-independent host cell signaling
Non-phosphorylated CagA have a different way of exerting effects within the cell. CagA translocation without phosphorylation leads to aberrant catenin activation, apical-junctional complex disruption and cellular polarity loss[84-89]. Relation between non-phosphorylated CagA and epithelial tight junction scaffolding proteins, zonula occludens 1 and junctional adhesion molecule A, results in imperfect association of tight junctions at located sites of bacterial attachment. Additional molecules includes E-cadherin, hepatocyte growth factor receptor c-Met, phospholipase C gamma (PL), adaptor protein Grb2, and kinase partitioning defective 1b/microtubule affinity-regulating kinase 2 (PAR1b/MARK2) resulting in mitogenic responses, interruption of cell-cell junctions and cell polarity destruction[84,87,88,90] (Figure 3). Recent study revealed that CagA directly binds to the cell polarity regulator such as PAR1b/MARK2. This binding prevents kinase PAR1b/MARK2 activity and deregulates the formation of mitotic spindle by cells which affects cell polarity[88,91].
Studies on transgenic mice revealed the correlation between CagA and oncogenesis by showing that CagA expression led to gastric epithelial cell proliferation and neoplastic changes. However, the following modifications were not detected in mice expressing phosphorylation-resistant CagA[52].
Presence of contradictory documentation on functionality of CagA as a bacterial oncoprotein in mammals exists besides solid proof provided by animal models. Pathological alterations described for transgenic CagA mice followed by absence of inflammation, which reflects disparity to what is seen in humans[52]. Although CagA act as oncoprotein, it remains to be explored why only few individuals inhabited by CagA-positive H. pylori develop GC. Recent study demonstrate that H. pylori prompts the presence of a host phospholipid, phosphatidylserine where CagA can explicitly interact and gain entry into the cells[92]. Focus of future research should be to define the exact mechanism of CagA internalization in gastric epithelial, factor responsible for regulation of this process and when during chronic infection CagA delivery in human epithelial cells.
VacA
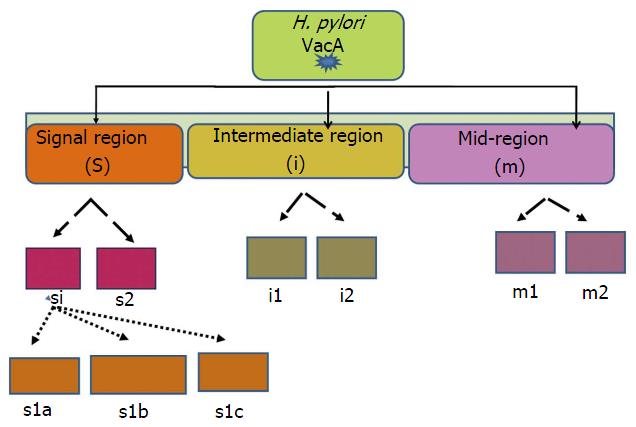
Another important H. pylori virulence gene is vacuolating cytotoxin (VacA), which encodes a bacterial toxin (VacA) that induces series of cascades leading apoptosis of epithelial cells via induction of cytoplasmic vacuoles (Figure 4). VacA is found throughout the H. pylori strains. The diverse polymorphic form of VacA are related with clinical outcomes[93]. Considerable genetic variations are found in: The s (signal) region with alleles s1a, slb, slc, or s2; the m (middle) region with m1 or m2 alleles; and the i (intermediate) region with type i1 or i2 alleles (Figure 4).
Figure 4 Representation of Helicobaceter pylori major virulence factor, vacuolating cytotoxin containing three domain 1: signal sequence(S) 2: middle region (m) 3: recently identified intermediate region (i) s, m and i region are further stratified into the subtypes s1, s2, m1, m2 and i1, i2 respectively.
TLR4: Toll like receptor 4; T4SS: Type IV secretion system; VacA: Vacuolating cytotoxin; CagPAI: Cag pathogenicity island; GC: Gastric cancer; ROS: Reactive oxygen species.
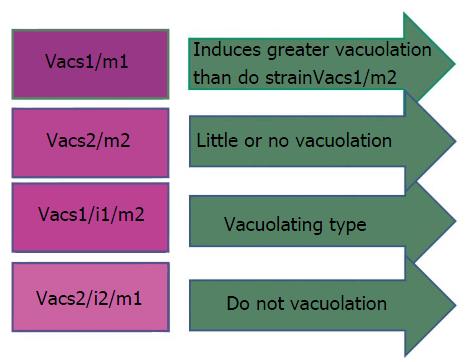
H. pylori strains having combination VacAs1/m1 or vacAs1/m1/i1 are associated with increased risk of progression to premalignant lesion and GC than vacA s2/m2 or vacAs2/m2/i2 strains[94] (Figure 5).
Figure 5 Vacuolating cytotoxin of Helicobacter pylori may have any combination of signal sequence and mid region with different virulence activities as stated above.
Vac: Vacuolating cytotoxin.
OTHER RISK FACTORS
Besides H. pylori, the following other environmental factors are considered to contribute in the pathogenesis of GC.
Diet
The variations in GC incidence are due to environmental inputs, particular in dietary pattern. Previous accumulating studies have been indicated that downward trend in GC occurrence. This may be due to the advent of widespread refrigeration of foodstuff and reduction in dependency on food preservation. In addition, other studies have suggested that a preventive role of diet containing fresh vegetables and fruits. However, data from European prospective study failed to show an overall association between fresh fruits and vegetables intake and GC risk[95]. Recent studies being conducted on this field revealed that a significant association between total dietary vegetables contents (onion and garlic intake) and intestinal GC subtypes.
Additional studies are required for demonstration of positive association between H. pylori eradication and prevention of cancer. The controversy related to point of no return in case of atrophy and metaplasia is still debatable. Proposed studies on side effects and expenses of such preventive measures are required in future for proper management and treatment of GC, therefore GC prevention remains a key part of research on H. pylori.
Salt
H. pylori is not the only the culprit for the development of GC; other influential causes include host polymorphisms and environmental elements (Figure 2). High dietary salt intake was found to be uniformly been associated with an increased risk of GC[12,96]. Two studies, one study from Japan and other a case-control study from South Korea stated that H. pylori-infected subjects taking high-salt diet had an greater risk of GC than those with lower levels of salt[97,98]. Association between the frequency of H. pylori infection and amount of dietary salt intake is reported in another study[99].
Research on Mongolian gerbils had shown that the H. pylori presence and usage of a more salt containing diet applied concerted effects on development of precancerous satge[100,101]. Additional study on H. pylori-infected gerbils demonstrates that there is a positive association between level of severity of gastric inflammation and rate of proliferation of epithelial cells in gerbils consuming high-salt diet than those consuming a normal diet[100]. Similar studies on gerbils infected with H. pylori, when treated with carcinogen (N-methyl-N-nitroso urea) shows that higher frequency of GC related with animals consuming high-salt diet as compared to animals with a normal diet[101,102].
Mechanisms behind the high-salt diet increases the risk of development of GC in humans remains unclear. Among various explanations, one plausible hypothesis is that salt may lower the threshold for malignant transformation by altering the physiology of gastric epithelium thus allowing entry of carcinogens into gastric tissue and resulting in damage to gastric mucosa. Another possibility is that high salt intake might be regulating the gene expression in H. pylori. Two independent studies suggested that consumption of excess amount of salts in diet leads to higher expression of H. pylori virulence factors[103,104].
Dietary antioxidants
Many studies had proved the antioxidants present in food in green vegetables and fruits plays a preventive role against progression of GC[105]. There is scarcity on studies on association of H. pylori infection with nutritive elements in gastric carcinoma. A case-control study recommended that consistent excessive consumption of vitamin C and carotene might be able to curtail the casual for developing GC in subjects having infection of H. pylori[106].
A randomized study on population susceptible for GC development demonstrated that combination of vitamin C and carotene dietary supplements and H. pylori eradication increases the preneoplastic lesions regression at 6 years of follow-up; at another 6 years follow-up lacking dietary supplements, the protective role of vitamin C and carotene gradually end up[7]. These results were also validated by other studies[95,106]. A similar study from Hawaii proof that consumption of fresh vegetable among H. pylori infected individuals provided a little protection against GC occurrence[107]. On the contrary, other studies fail to provide a positive association between H. pylori infection and plasma vitamin C level, with risk of GC incidence[108]. Additional research is required to determine whether antioxidants are capable of providing protection against GC among H. pylori infected patients.
Cigarette smoking
It is evident from various studies that cigarette smoking is associated with risk of developing GC in H. pylori infected subjects. In Japan, cigarette smoking and H. pylori infection together are considered as potential threat for developing GC[109]. Swedish and German population-based case control studies also demonstrated combination of cigarette smoking and infection by CagA positive H pylori strains increased the risk of developing GC (Figure 2). Los Angeles study also reported a tendency toward increased risk of GC in smokers[59,110]. On collectively analyzing studies, it emerges that there exists relationship between H. pylori infection and smoking with increased risk of developing GC.
Helminth infection
H. pylori co-infection with helminths may have some impact in disease pathogenesis. Reduced Th1 response associated with higher levels of Th2 cytokines was reported in one study[111]. Another study on Colombian children from a coastal region having infection of both helminths and H. pylori, showed a higher Th2 associated IgG1 response[112]. Further studies are needed to assess the impact of H. pylori and helminthes co-infection in disease pathogenesis.
FOCUS OF FUTURE ENDEAVORS
Gastric cancer remains a major threat to mankind. Improvement in living standards, increase awareness in sanitation and hygiene practices, reduction in intake of salted food products and advent of refrigeration in households resulted in measurable decline both in incidence of H. pylori infection and GC. Although both H. pylori infection and GC are showing decreasing trends in the developed world, they still remain a major threat to human population in the developing countries. Therefore, there is a need for improvement in early diagnosis, identification of risk factors, and development of preventive strategies and initiation of timely therapeutic interventions, especially focused for the developing countries. Further, it remains to be investigated why a small fraction of individuals colonized by H. pylori develop GC, and future research should focus on bacterial, host genetics, environmental and dietary factors.
There is need to formulate clear cut recommendation for screening and timely intervention of high risk population with family history of GC. Whether all high-risk areas should undergo routine screening of H. pylori infection is still questionable. Since the patients having atrophic gastritis or dysplasia in the gastric mucosa are at increased risk of developing GC, there is a need for special recommendations including endoscopic surveillance for such patients.
ACKNOWLEDGMENTS
Jahanarah Khatoon and Ravi Prakash Rai gratefully acknowledge Indian Council of Medical Sciences, for junior research fellowship through Fellowship sanction No. 3/1/3/3 JRF-2011/HRD-87(32606) and 3/1/3/JRF-2012/HRD-162 (80220) respectively.
P- Reviewer: Guimaraes NM S- Editor: Song XX L- Editor: A E- Editor: Li D