INTRODUCTION
Heavy alcohol consumption has classically been associated with an increased risk of bone fractures. In 1977, Oppenheim[1] described the “battered alcoholic syndrome” in patients with three or more fractures in different stages of healing. In that study, Oppenheim suggests that alcohol itself has direct negative effects on bone metabolism. In fact, other authors have found a similarity between alcoholic patients and the postmenopausal and geriatric population due to an increased incidence of fractures and a lower bone density. In 1965, Saville[2] described that the bone density measured in the left iliac crest of alcoholics below 45 years of age was similar to that of non-alcoholic men and women older than 70 years. Snell[3] described in 1971 that the pattern of fractures seen in alcoholics is similar to that seen in patients 20 years older. And in a classic study published in 1980, Israel et al[4] found an increased prevalence of rib and vertebral fractures in alcoholic patients and therefore suggest using routine chest X rays to identify problem drinking and to screen the general population for alcoholism.
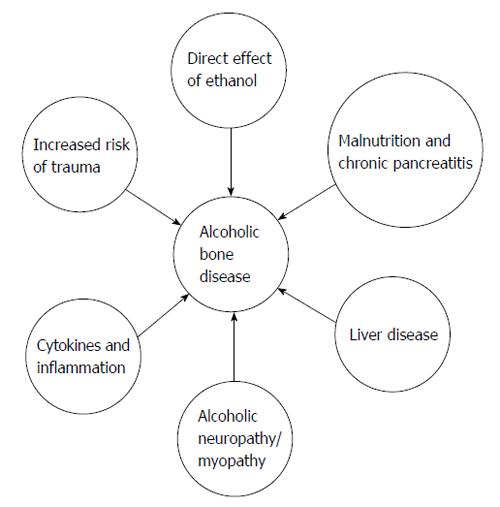
Although alcoholics may have an increased risk of trauma and subsequent fractures, alcohol itself has a negative impact on bone metabolism. Alcoholism has been associated with growth impairment, osteoporosis, osteomalacia, fractures, delayed fracture healing, and aseptic necrosis (primarily necrosis of the femoral head)[5,6]. The mechanisms that explain the loss of structural integrity of bone are partially known. These include a direct toxic effect of ethanol on bone synthesis and systemic alterations such as malnutrition and malabsorption, liver disease, altered hormonal and cytokine profiles, alcoholic myopathy, and neuropathy (Figure 1)[6]. In this review we will outline each of these mechanisms.
Figure 1 Factors that contribute to bone disease in alcoholic patients.
ALTERED BONE SYNTHESIS. DIRECT EFFECTS OF ETHANOL
As mentioned above, in 1965 Saville[2] had already described decreased bone density among alcoholic patients. In 1989 Diamond et al[7] found that drinkers had significantly less osteoblastic activity than teetotallers and suggested that reduced bone formation and mineralization is due to osteoblastic dysfunction. This has been reproduced in a study by Gonzalez-Calvín et al[8] where they found that chronic ethanol consumption reduces bone mineral density regardless of liver cirrhosis. In recent decades osteocalcin, a polypeptide bone matrix protein also known as bone gamma-carboxyglutamic acid-containing protein or BGLAP, has been described as a specific marker of osteoblast function and bone synthesis[9]. In the same study by Diamond et al[7], they described decreased osteocalcin levels in alcoholics. The same finding was also described in a more recent study by Santori et al[10] in 2008.
Giuliani et al[11] found that in murine bone marrow cultures, ethanol and acetaldehyde (the main metabolite of ethanol) significantly reduced osteoblast proliferation and decreased the number of colony forming units for fibroblasts. This may explain the decreased bone mineral density (BMD) in alcoholics. In fact, Torricelli et al[12] found in an in vitro model that osteoblast proliferation is affected more significantly by exposure to alcohol than by estrogen deficiency. Therefore, chronic alcoholism may have even more deleterious effects on bone metabolism than menopause.
These findings contrast with those described by Lau et al[13] in which they found that ethanol had a dose-dependent effect on bone formation. These authors found that low levels of alcohol increased bone proliferation but that alcohol might interfere with the normal bone remodeling process[13]. Similar results were reported by Marrone et al[14] where postmenopausal moderate alcohol consumers were found to have increased BMD and reduced bone turnover markers. However, in several studies the amount of alcohol consumed and the presence of osteopenia adopts a J-shaped curve where heavy drinking clearly has detrimental effects on BMD and the risk of fractures[15,16].
Other studies have also suggested that the inhibition of bone formation in chronic alcoholism may be mediated by the accumulation of reactive oxygen species (ROS). Chen et al[17] found that in a rat model ethanol up-regulated NADPH oxidase expression in osteoblasts. Administration of antioxidants such as N-acetyl cysteine prevented bone loss in rats exposed to ethanol[17]. This was also shown in a study by Lee et al[18] and Choi et al[19] where an NADPH oxidase inhibitor called apocynin protected osteoblasts from oxidative damage and increased cell survival, calcium deposition and osteoprotegerin release. Consistent with these findings, Chen et al[20] showed that ethanol-induced TNF-α secretion also promotes differentiation of mesenchymal cells towards adipose tissue. All of these effects are in line with the observation performed by Maurel et al[21] regarding osteocyte apoptosis suffered by osteocytes exposed to ethanol.
However, the changes in bone metabolism are not permanent and bone disorders in alcoholic patients have been considered by some authors as reversible[22,23]. In fact, in the study mentioned above by Gonzalez-Calvin they found that after just 7 d of abstinence, osteocalcin levels increased significantly[8]. We found similar results among 60 patients who were followed-up after admission and it was found that those who had stopped drinking showed an increase in total BMD as measured by bone densitometry[24].
While there are several studies that describe the effect of alcohol on bone metabolism in adults, adolescence is an important period in determining bone mass. In a study performed in rats by Turner et al[25] in 1988, it was found that tibial length was lower in animals exposed to ethanol and that there was an increase in resorption and a loss of trabecular bone. While bone disorders associated with alcoholism have been reported to be reversible in adults, osteopenia was shown to not be completely reversible in growing Sprague-Dawley rats exposed to ethanol[26]. We found among two sets of patients that those who began to drink before 18 years of age were shorter than controls. We also found that these patients had Harris lines in their right tibiae, a condition associated with episodes of stunted growth and subsequent recovery[27].
ALTERED HORMONE LEVELS MUSCLE ATROPHY
The effects of alcohol administration have been described in rats where it resulted in the stimulation of adrenal steroid production that did not occur when rats were hypophysectomised[28]. Smith[28] found that stimulation of the adrenal cortex led to increased excretion of calcium in urine which may contribute to bone demineralization in alcoholics. In a study by Badrick et al[29] a 3% increase in cortisol levels per unit of alcohol consumed was found. They also showed a reduced control of the hypothalamic-pituitary-adrenal (HPA) axis. However, what is usually described among alcoholics is a Pseudo-Cushing’s syndrome in which it has been suggested that there is increased secretion of corticotropin releasing hormone with a normal HPA axis[30-33]. While these patients usually have physical signs of hypercortisolism, they can be differentiated because Cushing’s syndrome is associated with a higher cortisol midnight:morning ratio, higher midnight serum cortisol levels, and higher levels of corticotropin-releasing hormone/stimulated cortisol after dexamethasone suppression[34]. While cortisol may theoretically reduce BMD, we have failed to find a relationship between cortisol and BMD in several studies[27].
Muscle mass and strength are determinants of bone mass and muscle atrophy and bone loss have already been described as interdependent. In a study by Lloyd it has been found that in mice subjected to mechanical unloading, muscle atrophy precedes bone loss and contributes to trabecular bone loss[35]. In this sense, muscle atrophy associated with alcoholism is yet another factor that contributes to decreased BMD in alcoholics. Muscle atrophy in alcoholics is, in turn, associated with vitamin D deficiency and low testosterone levels.
Decreased vitamin D levels have been described in patients with chronic liver disease[36]. In a study by Fisher et al[37] it was found that cirrhotic patients had a higher prevalence of vitamin D deficiency (defined as levels below 50 nmol/L) and that these levels were significantly lower in Child-Pugh class C patients. A correlation between serum vitamin D levels and INR and albumin was also found. In addition, vitamin D has an important effect on muscle and a specific vitamin D receptor has already been described in muscle fibers[38-40].
On the other hand, hypogonadism has been classically described in men with chronic alcoholism and this has been attributed to a defect in testicular androgen production or to defects in luteinizing hormone secretion[41]. Testosterone increases muscle mass so that testosterone deficiency in alcoholics contributes to the muscle atrophy that is seen in these patients.
Muscle function affects bone through the activation of the Wnt/β-catenin pathway mediated by the load exerted by muscle contraction. The Wnt signaling cascade was described in the 1980s and its role in bone formation has been extensively studied in recent years[42]. This cascade regulates osteogenesis through the repression of mesenchymal differentiation towards adipocytes and chondrocytes, favoring osteoblast differentiation while also blocking osteoblast apoptosis[42]. In a study by Kramer et al[43] it was shown that β-catenin deficient mice had a low bone mass associated with increased osteoclast activity. Independently on the effects of muscle mass, Chen et al[44] described that ethanol inhibits the Wnt1/β-catenin signaling pathway, leading to a shift of differentiation of bone marrow cells towards adipogenesis instead towards osteoblasts. Li et al[45] have shown that sclerostin could antagonize Wnt signaling by binding to low-density lipoprotein receptor-related proteins 5 and 6 (LRP5 and LRP6). In this sense, loss of sclerostin should be associated with an increased bone mass but low sclerostin levels have been described in osteoporotic women[46]. In a preliminary study on alcoholic patients we have found increased levels of sclerostin which could explain decreased BMD[47].
INCREASED BREAKDOWN THE ROLE OF CYTOKINES AND THE OPG/RANK-L SYSTEM
Bone resorption depends on osteoclasts and osteoclast differentiation is in turn mediated through the receptor activator of nuclear factor-kappaB (RANK)/RANK ligand (RANKL)/osteoprotegerin (OPG) signaling pathway which was described in the late 1990s[48]. This pathway regulates the formation and activation of osteoclasts. On the other hand, OPG binds to RANKL and prevents it from binding to RANK. Osteoprotegerin was first described by Simonet et al[49] in 1997 as a novel member of the TNF receptor superfamily that is able to block osteoclast formation. Therefore, the OPG/RANKL ratio is an important marker of bone formation/degradation and is also regulated by the Wnt/bcatenin canonical pathway. This pathway up-regulates OPG expression and down-regulates RANKL expression, thus suppressing osteoclastogenesis[50].
Hofbauer et al[51] report that proinflammatory cytokines such as interleukin (IL)-1beta and tumor necrosis factor (TNF)-α but not IL-6 increase osteoprotegerin ligand levels (whose effects are prevented by its receptor, osteoprotegerin) which may stimulate the formation of osteoclasts. This may explain reduced BMD among alcoholics in which there are increased levels of TNF and IL-6 due to the effect of activated Kuppfer cells mediated by gut-derived endotoxin[52,53]. Proinflammatory cytokine levels are also increased in alcoholics due to associated infections that are common among these patients. Therefore, the effects of increased TNF-α levels involve both decreased osteoblast differentiation and increased bone resorption. In accordance with the resorptive effect of TNF-α (a molecule which induces increased ROS production), NADPH oxidase derived ROS are also involved in increased osteoclastogenesis[54].
However, despite these experimental evidence, in alcoholic patients the effects of ethanol on osteoclast activity is less clear[55] and while some authors report increased bone turnover in alcoholic cirrhosis, others find no differences with controls. In one study we found increased levels of OPG in alcoholic patients which may be interpreted as a compensatory mechanism in the context of osteopenia[56].
MALNUTRITION AND VITAMIN D DEFICIENCY IN ALCOHOLICS
Malnutrition is one of the main factors that contributes to decreased bone mass among alcoholic patients and is associated with decreased caloric intake, impaired absorption, and decreased hepatic synthesis of proteins. Alcoholism-associated malnutrition is multifactorial and is related to irregular feeding habits, cirrhosis with ascites, high ethanol intake and social factors[57]. Interestingly, we have shown that low BMD is not related to the intensity of alcoholism or calciotropic hormone levels but to malnutrition[27].
Specific nutritional deficiencies such as lower zinc levels have also been described among chronic alcoholics[58]. Zinc deficiency in these patients may be due to impaired absorption, increased urinary excretion or decreased affinity of albumin for zinc[59]. As described by Yamaguchi[60], zinc stimulates osteoblastic bone formation and mineralization while its deficiency is associated with decreased bone mass. This suggests a possible role of zinc supplementation in the prevention of osteoporosis in alcoholic patients.
Bone changes in alcoholic patients are also related to deranged vitamin D metabolism. As reviewed previously by Pitts et al[61], low vitamin D levels in alcoholics are due to reduced hepatic 25-hydroxylase activity, malabsorption, irregular feeding habits and lack of sun exposure. Low vitamin D levels also have an effect on calcium and phosphate homeostasis due to malabsorption of calcium and resultant hypocalcemia. Laitinen et al[62] described in 1991 that moderate drinking elevates parathyroid hormone (PTH) levels while acute ingestion of alcohol leads to a transient decrease in PTH. This was reproduced in an animal model studied by Chen et al[63] where it was found that long term treatment of hamsters with ethanol lead to stimulation of the secretory activity of the parathyroid gland. Another effect of ethanol on calcium homeostasis was described by Kalbfleisch et al[64] in 1965 when it was described that the administration of ethanol increased urinary excretion of calcium and magnesium.
POTENTIAL THERAPIES FOR ALCOHOL-INDUCED BONE DISEASE
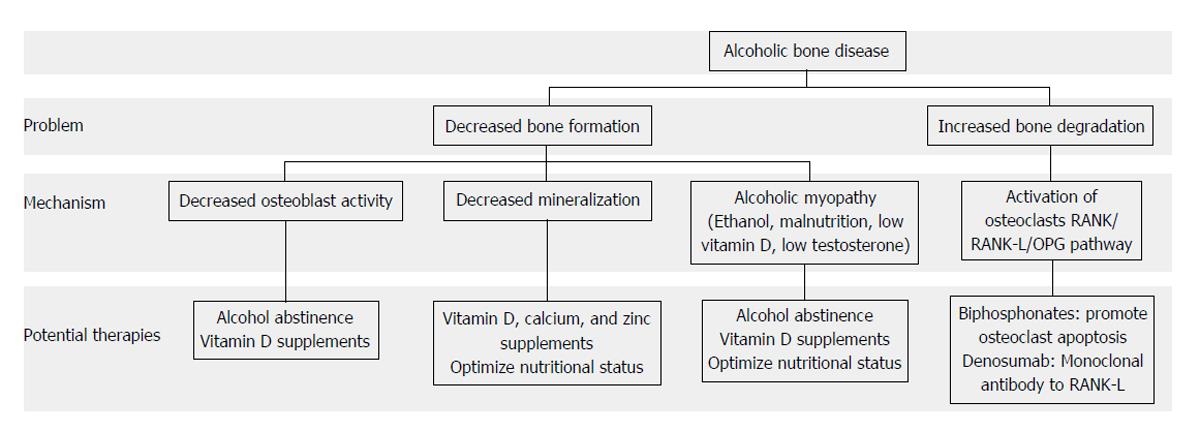
As illustrated throughout this review, alcohol has a toxic effect on bone mediated by decreased bone formation and increased bone degradation that are, in turn, influenced by nutritional, hormonal, and proinflammatory factors. Therefore, the treatment of osteoporosis in alcoholic patients should be aimed towards increasing osteoblastic and decreasing osteoclastic activity (Figure 2). As mentioned above, the supplementation of alcoholic patients with micronutrients such as zinc may also have an important role in the prevention of bone disease. In this sense, we have shown that in Sprague-Dawley rats zinc supplementation increases osteoid area but randomized trials in patients are still needed[58].
Figure 2 Pathogenesis of alcoholic bone disease and potential therapies.
Bone disease in alcoholic patients is due basically to decreased bone formation (decreased osteoblast activity and poor mineralization) and increased bone degradation (increased osteoclast activity). Potential therapies should counteract these effects but the mainstay of treatment should be alcohol abstinence. RANK: Receptor activator of nuclear factor kappa-B; RANK-L: RANK ligand; OPG: Osteoprotegerin.
Some studies have shown that vitamin D supplementation in mice exposed to ethanol may have beneficial effects on bone metabolism[65] and this has also been shown in animal models of acute alcohol consumption (equivalent to binge drinking)[66]. Once again, further studies in humans are needed to determine the impact of vitamin D supplementation and treatment with biphosphonates among alcoholic patients.
Biphosphonates inhibit bone resorption and have been widely used to treat osteoporosis in postmenopausal women. They promote the apoptosis of osteoclasts through the inhibition of farnesyl pyrophosphate synthase, an enzyme that is involved in the production of isoprenoid lipids. This impairs the isoprenylation of proteins that are involved in the survival of osteoclasts[67]. In a meta-analysis that studied 11 trials performed on postmenopausal women receiving alendronate, it was found that it reduced the risk of both vertebral and non vertebral fractures. It also increased bone density and this was dose and time dependent[68].
Biphosphonates have also been studied in patients with chronic liver disease. In a recent Cochrane meta-analysis it was found that treatment of patients with primary biliary cirrhosis with biphosphonates (etidronate or alendronate) did not decrease the risk of fractures compared with those who received placebo. They also had no significant effect on bone mineral density compared with placebo. In the trials analyzed they found that biphosphonates reduced the concentration of urinary amino telopeptides of collagen and serum osteocalcin[69]. Despite these findings, biphosphonates are well tolerated and are considered the mainstay of treatment in bone disease associated with both cholestatic and parenchymal liver disease.
Other classes of drugs that have been shown to be effective in postmenopausal osteoporosis and could theoretically benefit alcohol-induced osteoporosis are selective estrogen receptor modulators such as raloxifene and molecules that inhibit the activation of the RANK/RANKL pathway such as Denosumab. Raloxifene was studied in postmenopausal women in the MORE trial and it was found that continued therapy reduced the incidence of vertebral fractures, preserved bone mineral density and lowered markers of bone turnover[70]. On the other hand, Denosumab is a human monoclonal antibody to the RANKL, thus inhibiting its binding to RANK and decreasing osteoclast activity and subsequent bone resorption. Denosumab reduced the risk of vertebral, non vertebral, and hip fractures in postmenopausal women compared to placebo[71]. Once again, these results could be extrapolated to alcoholic patients.
P- Reviewer: Albuquerque A, Romani A, Yin H, Zhang XC
S- Editor: Gong XM L- Editor: A E- Editor: Liu SQ